Erythrocytes
Erythrocytes
Erythrocyte product focus
As well as supplying a wide range of blood group system antibodies, ARP also provides a variety of antibodies to important erythrocyte markers and cytoskeletal proteins. Red blood cell erythrocyte cell membrane proteins help maintain erythrocyte function while changing cell shape. Indeed, the ability of erythrocytes to change shape (deformability) under force without rupture (hemolysis) is a key characteristic in maintaining blood function [1].
The structure of red blood cells, specifically the cell membrane is complex and helps to maintain its biconcave shape. It is formed of a lipid bilayer with at least 20 major and over 850 minor proteins embedded within it. Band 3 (CD233) is the most abundant protein in the erythrocyte cell membrane. It plays a pivotal role in gas exchange and anchors to the cytoskeleton to help maintain the structure of red blood cells. Band 3 is typically found in complex with other proteins and 3 macrocomplexes have been described - an ankyrin complex, an actin junctional complex or as unbound band 3.
Other key proteins within the membrane and cytoskeleton include protein 4.1, protein 4.2, ankyrin, alpha spectrin, beta spectrin, actin and many members of the blood group antigen systems.
Defects in erythrocyte cell membrane proteins can result in severe clinical presentation [2]. For example, one of the most common causes of inherited hemolytic anemia is caused by hereditary spherocytosis (HS). HS results from the deficiency or dysfunction in genes coding for erythrocyte cell membrane or cytoskeletal proteins like protein 4.1, protein 4.2, band 3 (CD233) and alpha and beta spectrins. HS symptoms include anaemia, jaundice, fatigue and enlarged spleen (splenomegaly) and is typically diagnosed soon after birth.
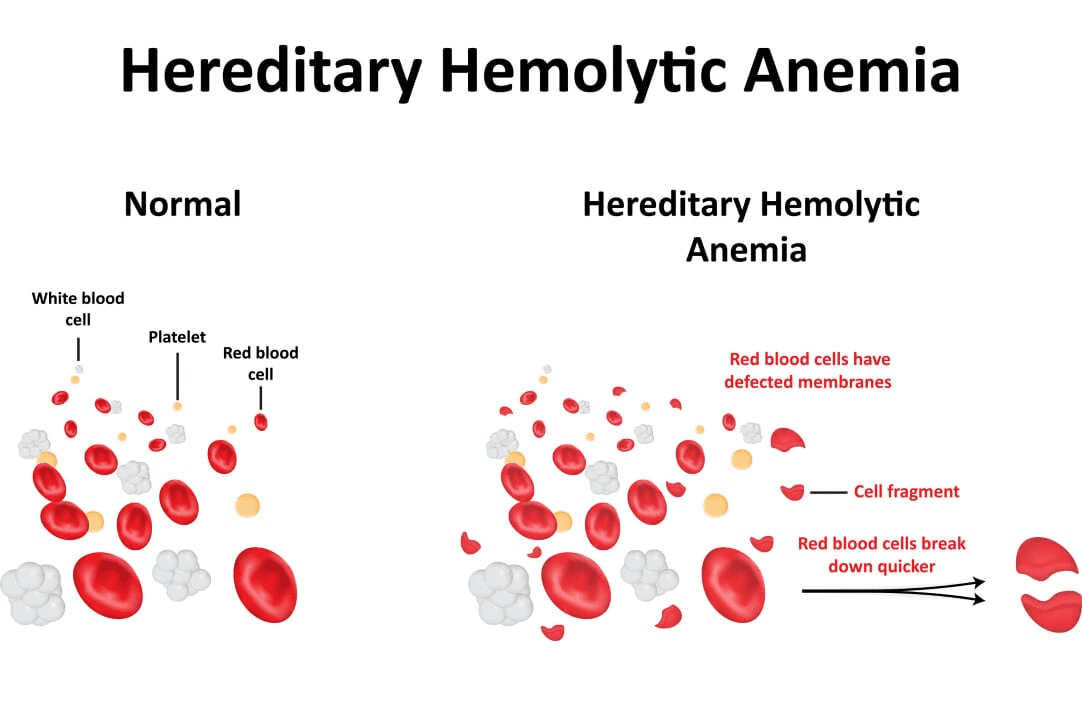
References
- Andolfo, I et al. New insights on hereditary erythrocyte membrane defects. Haematologica. 2016 Nov; 101(11): 1284–1294. doi: 10.3324/haematol.2016.142463
- Farias MG. Advances in laboratory diagnosis of hereditary spherocytosis. Clin Chem Lab Med. 2017;55(7):944–948. doi:10.1515/cclm-2016-0738




